The ability to predict drug efficacy and patient response to medicines allowing for personalised drugs and targeted therapies that have a greater chance of success in treating many diseases and health conditions. This is certainly something to strive for. Such personalisation is beneficial to reduce the time and costs involved in drug and treatment programs that currently take no account of the likelihood of success for particular patient groups. Not all patients react to drugs in the same way, whether as a result of pre-disposition markers, genetics or lifestyles.
Therefore, treatment needs to move beyond one size fits all. Identifying drugs and medicines with a higher chance of success for each patient will mean individual treatment plans to give faster, better outcomes and the time spent on treatments that would fail for pre-determinable reasons are reduced. Time and costs are reduced for hospitals and doctors.
Most importantly, reducing time spent on ineffective or experimental treatments by pin-pointing drugs that offer increased survival chances for patients drives personalised medicine forward.
So, is it possible?
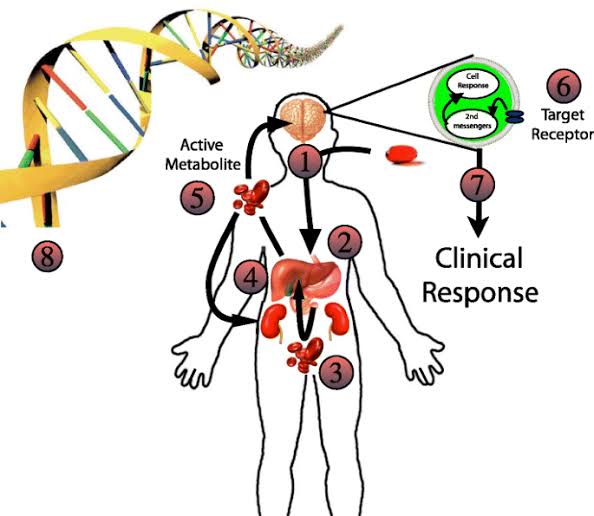
The power of Phosphoproteomics drives change. Specialising in cell signalling through interdisciplinary phosphoproteomics platforms to combine LC-MS/MS with proprietary bioinformatics enables bespoke research, development, and diagnostics. This capability allows for both clinical and pre-clinical identification of personalised medicine approaches. In addition, it is being used to monitor the efficacy of drugs on particular patients and groups. With the aim that both time and money to identify appropriate treatments is reduced.
How does it work?
A leading pioneer in precision medicine research and diagnostics tells us more about the work being carried out in this field. Kinomica has developed a phosphoproteomics platform known as KScan®, which can predict patient responses to targeted therapies. With an accuracy greater than existing state-of-the-art technologies or diagnostic testing. This technology supports drug discovery and development at both the pre-clinical stage and Phase I, Phase 2 or beyond.
Investigating modes of action and drug resistance mechanisms using KScan®’s proprietary computational algorithms to interrogate the phosphoproteomes and identify clinically precise biomarkers will significantly improve patient stratification.
It is a technology that enables the examination of over 10,000 proteins in one straightforward experiment. Researchers gain specific information on activated and deactivated proteins, which are highly relevant for personalised medicine.
The ability to carry out targeted testing and achieve fast, accurate results will save time, reduce costs, and provide critical information to improve patient outcomes.
Examples in action
The cell signalling processes that drive fibrosis and scarring in post-Covid lungs are being examined in a research project in conjunction with the Royal Brompton Hospital to identify whether specific kinase inhibitors that are already on the market to treat and prevent such fibrosis effectively. In this way, targeted therapies will be identified to provide personalised medicines to those in need.
It is also hoped that using KScan® will allow scientists and medical researchers at Kings College to look at the cell signalling processes in liver disease and liver cancer. Formalin-fixed and paraffin-embedded (FFPE) samples are being looked at as a potential tissue source for examining cell signalling and combined with a review of drugs typically used to treat diseased livers in patients. They seek to identify a correlation in cell signalling patterns to the drug responses for these patients. The outcome is expected to be a range of diagnostic tests that would improve precision medicines and personalisation for patients.
As far back as 2016, NHS England recognised the need to change from a blockbuster approach to drug development and patient treatment. The assumption that all patients respond similarly to drug therapies was not only false but costly too. The power of phosphoproteomics is making this a reality. With first-line treatments only being 30-60% effective, the benefit of phosphoproteomics to differentiate subtypes of patient reactivity to treatments creates an exciting new area of personalised medicine.